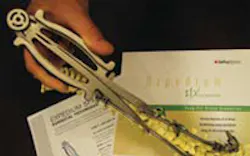
Direct metal laser-sintering (DMLS) technology has spawned a new foundation on how medical companies design components, Peter Ostiguy from DePuy Spine said at the first Rapid Implant Manufacturing Forum in Leuven, Belgium, in September.
Companies designed for manufacturability in the past, but with laser sintering, they now can design for functionality.
With direct metal laser-sintering, DePuy has reduced lead times in some projects by as much as 50 percent, even if product iterations became necessary. The company also predicts a wider use of direct metal lasersintering manufactured instruments to be used in operating theaters once EOS GmbH (www.eos.info), a maker of laser-sintering systems, launches its new hardenable EOS StainlessSteel PH1 material commercially.
EOS StainlessSteel PH1 fulfills ASTM E8 specifications as built, and complements the existing material EOS Stainless Steel 17-4. Pilot customers currently are testing PH1.
Other speakers at the forum reported the use of laser-sintered devices during medical operations. Such devices include acetabular cups and knee implants made from EOS CobaltChrome MP1.
An Italian company, ProtoCast, has developed a new manufacturing process for acetabular cups, which have already received CE certification.